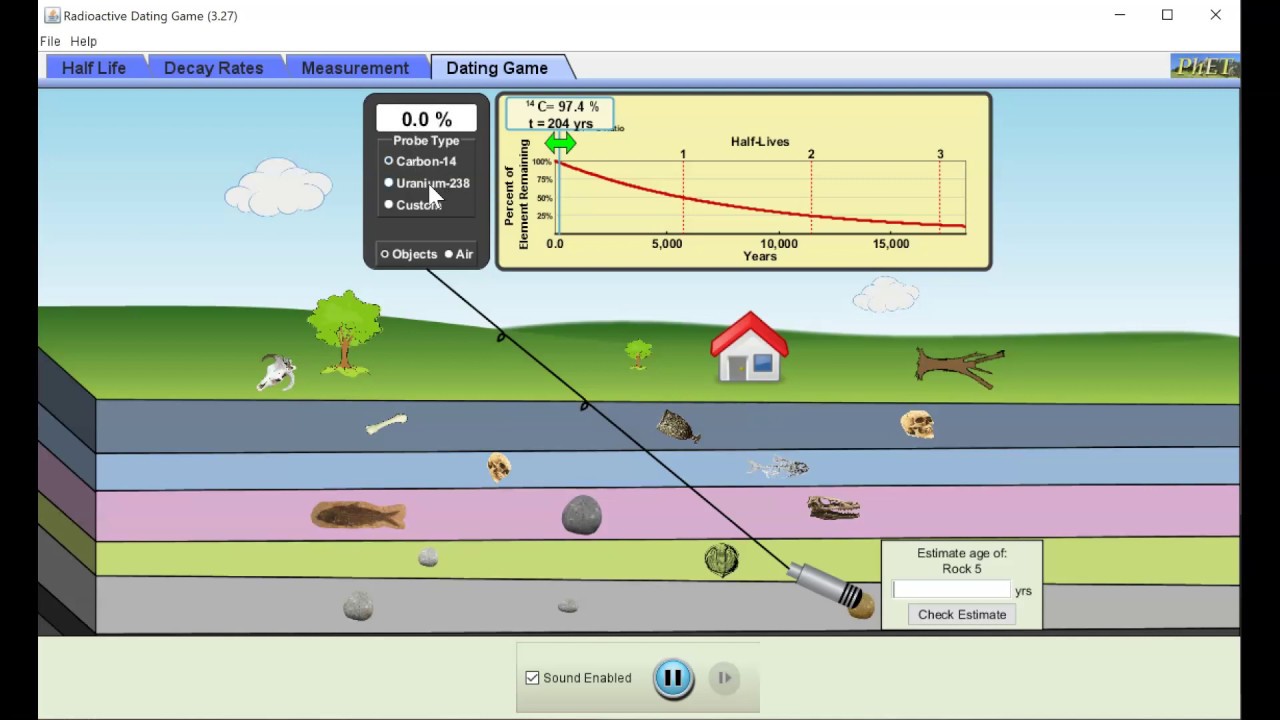
This video explains half-life in the context of radioactive decay. Radioactive decay occurs in atomic nuclei and therefore does not depend on chemical bonding or collisions of molecules. Carbon dating is a real-life example of a first-order reaction. 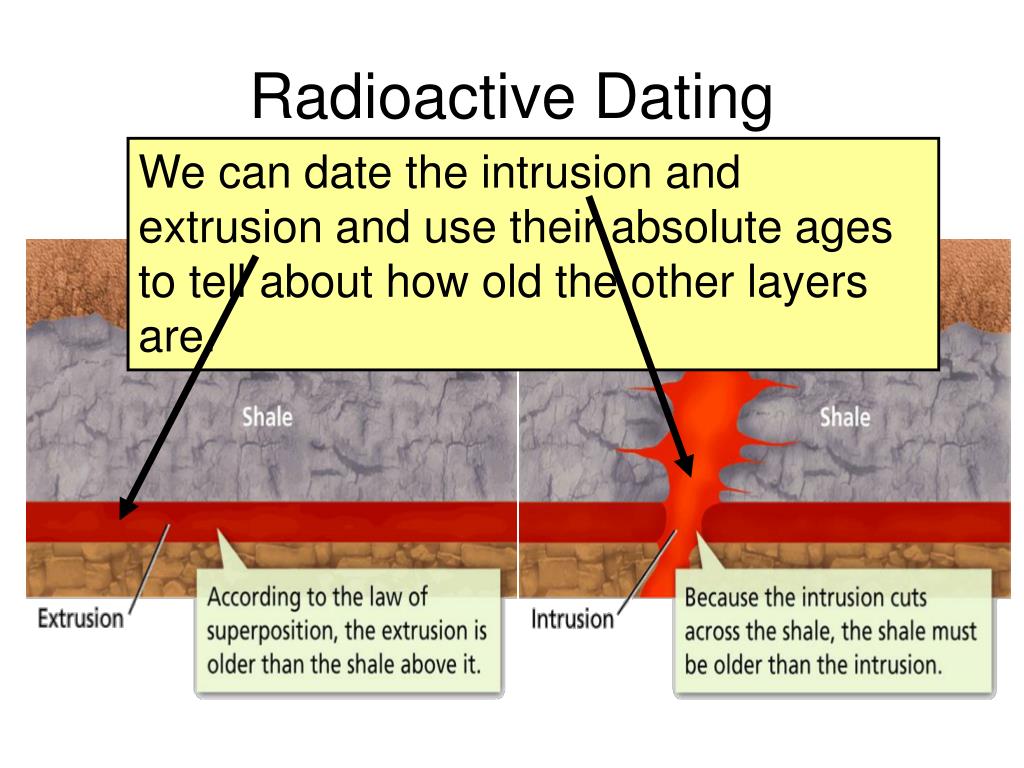
Consequently, unlike the situation with first-order reactions, the rate constant of a second-order reaction cannot be calculated directly from the half-life unless the initial concentration is known. The half-life increases as the reaction proceeds due to decreasing concentration of reactant. \nonumber \]įor a second-order reaction, t ½ is inversely proportional to the rate constant and the concentration of the reactant, and hence is not constant throughout the reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
